
Vagina
and Urethra
Anterior Repair and Kelly
Plication
Site Specific Posterior Repair
Sacrospinous
Ligament Suspension of the Vagina
Vaginal Repair of Enterocele
Vaginal Evisceration
Excision of
Transverse Vaginal Septum
Correction of
Double-Barreled Vagina
Incision
and Drainage of Pelvic Abscess via the Vaginal Route
Sacral Colpoplexy
Le Fort Operation
Vesicovaginal Fistula
Repair
Transposition
of Island Skin Flap for Repair of Vesicovaginal Fistula
McIndoe Vaginoplasty
for Neovagina
Rectovaginal Fistula
Repair
Reconstruction of
the Urethra
Marsupialization
of a Suburethral Diverticulum by the Spence Operation
Suburethral
Diverticulum via the Double-Breasted Closure Technique
Urethrovaginal
Fistula Repair via the Double-Breasted Closure Technique
Goebell-Stoeckel
Fascia Lata Sling Operation for Urinary Incontinence
Transection
of Goebell-Stoeckel Fascia Strap
Rectovaginal
Fistula Repair via Musset-Poitout-Noble Perineotomy
Sigmoid
Neovagina
Watkins Interposition Operation |
McIndoe Vaginoplasty for Neovagina
The McIndoe Vaginoplasty is indicated in patients with
congenital absence of the vagina, in patients whose vagina must be
removed, and in patients with severe stenosis following irradiation
therapy. A split-thickness skin graft has traditionally been utilized
with this operation, but we have changed to a full-thickness skin graft
over an expandable foam rubber mold placed in the canal between the
bladder and rectum. The use of the full-thickness skin graft reduces
the postoperative contraction previously noted with the split-thickness
skin graft requiring long-term utilization of a vaginal form, which
is undesirable if unnecessary. Recently, it has been shown that a full-thickness
skin graft takes as well as a split-thickness graft. Compared with
a split-thickness skin graft, a full-thickness graft allows sufficient
penetration of the transudate nutrients from the bed of the graft that
are necessary for nutrition during the first 72 hours until microcapillary
ingrowth has been completed. Full-thickness grafts do not have the
same degree of contraction as the split-thickness grafts.
Physiologic Changes. One of the unique
physiologic changes is the selection of thickness desired for the split-thickness
skin graft. The split-thickness graft of 1/12,000ths of an inch has
traditionally been used because it is thin enough to allow penetration
of the transudate nutrients from the bed of the graft that are necessary
for nutrition during the first 72 hours until microcapillary ingrowth
has been completed. A split-thickness graft of 1/12,000ths of an inch
is, however, associated with extensive contraction in the postoperative
period unless a vaginal form is worn for many months. Currently, there
has been greater utilization of thicker and thicker split-thickness
skin grafts until lately, full-thickness skin grafts have been utilized.
Initially, it was thought that the blood supply to these full-thickness
skin grafts would be insufficient and there would be excessive amounts
of necrosis. The copious blood supply of the pelvis has demonstrated,
however, that the full-thickness skin graft will survive. Thus with
excellent survival of a full-thickness skin graft the patient is spared
long-term vaginal form dilatation.
Points of Caution. Careful
dissection between the bladder and rectum is necessary to avoid cystotomy
and enterotomy. Meticulous hemostasis is essential for the graft
to adhere to the vaginal walls.
The skin grafts should be of uniform size and predominately of full-thickness.
The air-powered dermatome is preferred over the Reese or the electrical
dermatome because it gives a uniform specimen of consistent thickness.
The full-thickness grafts can be taken by the hand technique with a
standard scalpel. We now prefer to take these from the skin covering
the inguinal ligaments bilaterally. If there is tension on the suture
line closing these donor site wounds, the new SureClosure skin-stretching
system can be used to adequately close the wound. This has the advantage
of leaving the patient with a cosmetic scar over the inguinal ligament
versus a large scar over the buttocks or legs.
The vaginal mold should
be soft and flexible. Foam rubber confined to a rubber condom has produced
a soft vaginal mold that expands against the graft in a gentle manner
without producing points of pressure. Excessive pressure could produce
necrosis with possible vesicovaginal or rectovaginal fistula formation.
The
vaginal form should be left in place for a minimum of 10 days. The
form should then be removed, and the skin graft donor site should be
inspected.
One of the most important criteria for this operation
is the surgeon's conviction that the patient is mature enough to wear
the vaginal form for at least 6 months, unless she is engaging in regular
sexual intercourse. Failure to wear the vaginal form, especially with
the split-thickness skin graft, is the major cause of failure in this
operation. This generally occurs in adolescent girls who do not understand
the importance of keeping the vaginal form in place even if it is inconvenient
or uncomfortable.
Technique
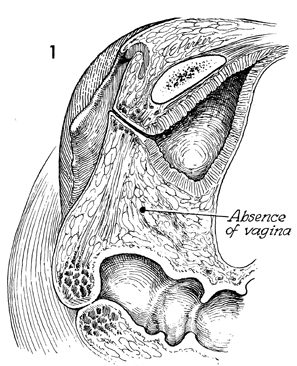
A sagittal section through the pelvis
demonstrates congenital absence of the vagina. The site of dissection
will be in the areolar tissue between the bladder and rectum.
The space is exaggerated here for illustration. The dissection
should continue cephalad to 1-2 cm before the level of the peritoneum.
Deeper dissection may allow the development of an enterocele. |
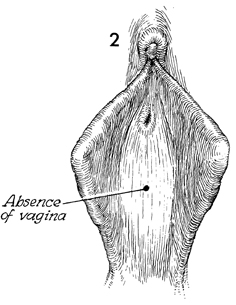
In this view of the introitus in
congenital absence of the vagina, a slight indentation or dimple
may exist where the urogenital sinus in the fetus failed to invaginate
and fuse with the Mullerian duct to develop a normal vagina. |
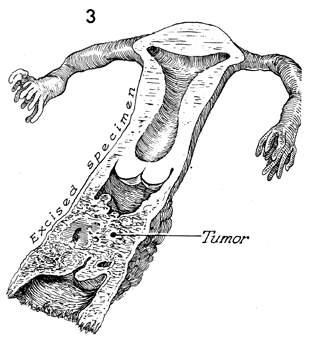
A second cause of absence of the
vagina is surgical removal for oncologic reasons. |
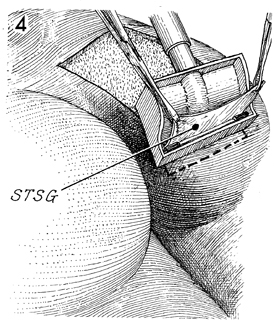
The air-powered dermatome is preferred
because of the uniform thickness and size of the graft produced.
The setting on the air-powered dermatome can be adjusted to 18/1,000ths
or 24/1,000ths of an inch. STSG identifies the split-thickness
skin graft. |
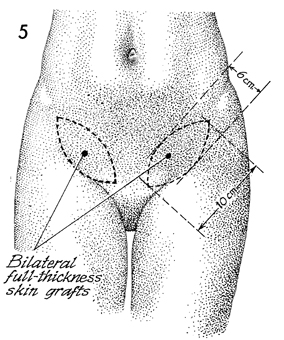
Two proposed paddle-shaped full-thickness
skin grafts over the inguinal ligaments from the anterior superior
iliac spine to the pubic tubercles are depicted. These paddles
should be approximately 10 cm in length and 6 cm in width. |
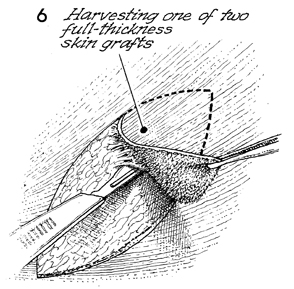
Harvesting of the full-thickness skin
graft is performed with the standard scalpel after marking the
shape of the paddle in ink on the skin.
|
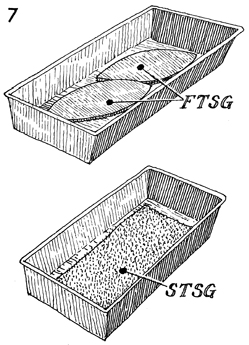
The full-thickness skin grafts (FTSG) are
placed in a graft pan and moistened with saline. The same is
true for the split-thickness skin graft (STSG) if that
is preferred to the full-thickness skin graft. |
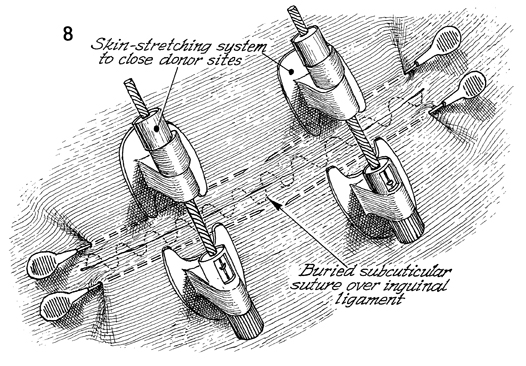
After harvesting the full-thickness skin
graft from the inguinal ligaments, if closure of the wound is
on tension, the new SureClosure skin-stretching system can be
utilized as shown. The stabilization needles are inserted in
the subcutaneous space parallel to the line of the incision.
Two, 4, or 6 of these can be utilized as necessary. Usually,
no more than 2 are required. The skin-stretching device is anchored
into the needle, and the ratchet on the skin-stretching device
is activated until the skin is stretched to close the incision.
Presently, a subcuticular suture of 3-0 delayed synthetic absorbable
suture is utilized to close the wound. |
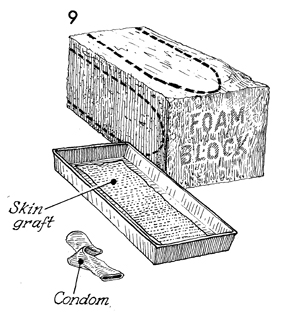
A block of ordinary foam rubber, such as
that used in the upholstery industry, is sterilized by gas autoclaving.
The shape of the proposed vaginal form is outlined with brilliant
green solution. The split-thickness skin graft is removed
from the adhesive tape of the dermatome and placed in a sterile
pan filled with saline solution. Ordinary contraceptive condoms
that have been sterilized by gas autoclaving are used. Generally,
two condoms are used to ensure against leakage of fluid into
the foam rubber form. |
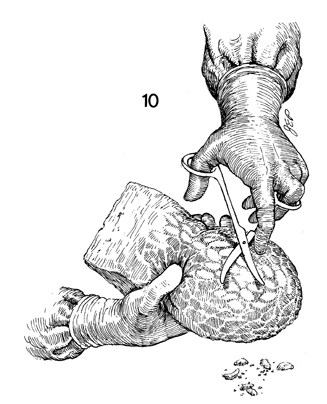
The foam rubber block is held in one hand
and, with curved Mayo scissors, is shaped into the desired vaginal
form. |
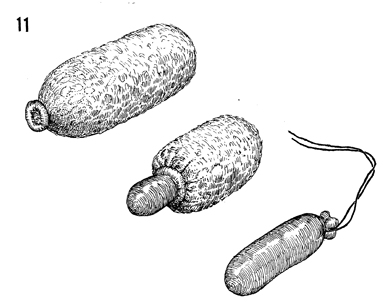
The condom is slipped over the foam rubber
vaginal form. The condom can be removed partially or completely
if the size of the vaginal form needs to be adjusted in diameter
or length. When the size of the vaginal form is satisfactory,
the end of the condom is tied with a 2-0 synthetic monofilament
permanent suture. |
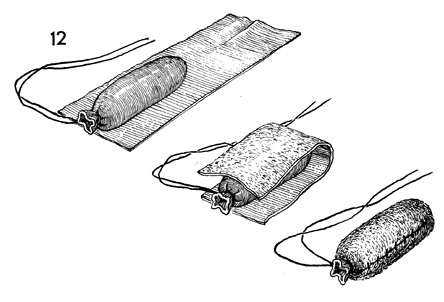
The vaginal form is laid on the epidermal
side of the split-thickness skin graft. The split-thickness skin
graft is folded over the vaginal form and sutured along its seam
with interrupted 4-0 synthetic absorbable suture. Excess graft
is trimmed away. |
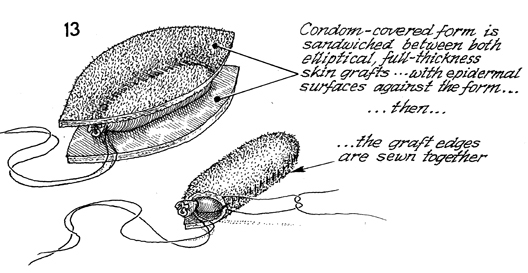
The full-thickness, paddle-shaped skin grafts
are placed over a foam rubber condom, and the margins are sutured
together with synthetic absorbable suture. |
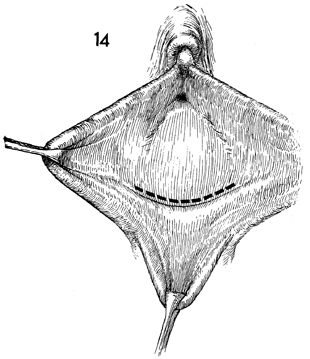
The dimple in the introital area is identified.
The labia are retracted with Allis clamps, and a transverse incision
is made in the epithelium. |
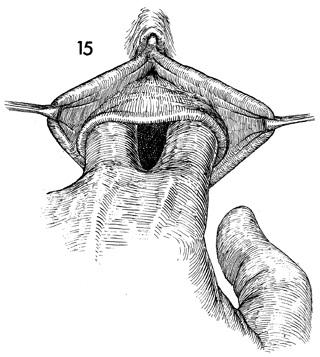
Blunt dissection with the fingers opens the
space between the bladder and rectum. Once the correct plane
is reached, i.e., below the pubovesical cervical fascia under
the bladder and superior to the perirectal fascia over the rectum,
gentle blunt dissection is all that is needed to create an adequate
cavity. |
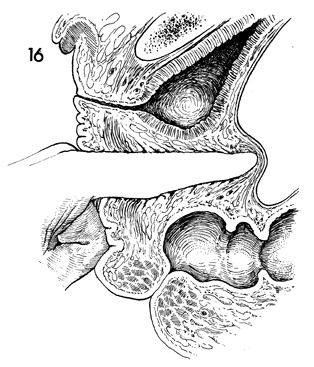
A sagittal section shows the dissection carried
approximately 2 cm from the peritoneum. This will reduce the
incidence of enterocele in these patients. |
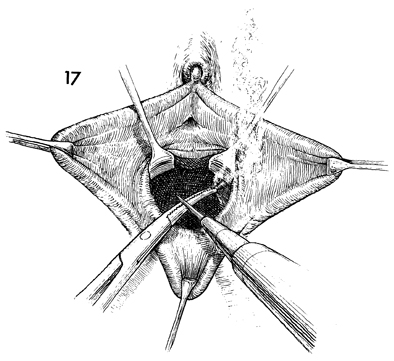
Meticulous hemostasis should be maintained
throughout the cavity. |
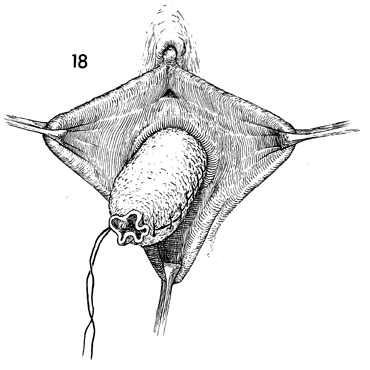
The skin-covered form is inserted into the
cavity. |
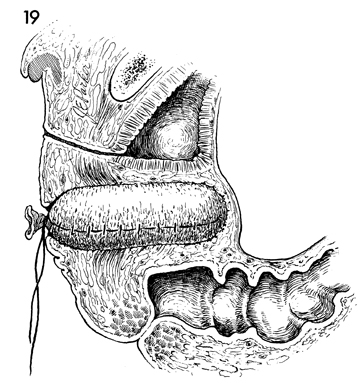
A sagittal view of the pelvis shows the skin-covered
form inserted into the new vaginal canal. |
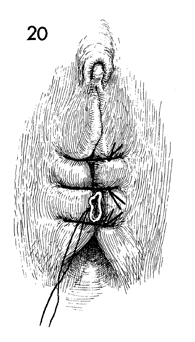
To hold the form in place for 12 days, the
labia are sutured in the midline with interrupted 0 nylon sutures
without tension. |
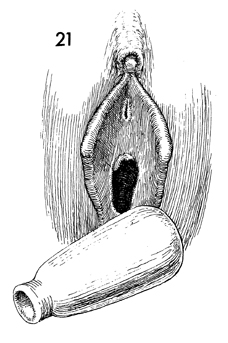
After the foam rubber vaginal form has been
removed on the 12th postoperative day, the new vagina should
be thoroughly inspected and irrigated with normal saline solution.
A permanent vaginal form made of soft rubber or silicone should
be fitted and inserted. Care should be taken that the new vaginal
form does not protrude beyond the introitus. Protrusion will
erode the introital area and cause pain, discouraging the patient
from continuing to use the form. |
|
|





















